Diagnostic Procedures in Respiratory Disease : Introduction
The diagnostic modalities available for assessing the patient with suspected or known respiratory system disease include imaging studies and techniques for acquiring biologic specimens, some of which involve direct visualization of part of the respiratory system. Methods to characterize the functional changes developing as a result of disease, including pulmonary function tests and measurements of gas exchange, are discussed in chapter Disturbances of Respiratory Functions.
Imaging Studies
Routine Radiography
Routine chest radiography, generally including both posteroanterior (PA) and lateral views, is an integral part of the diagnostic evaluation of diseases involving the pulmonary parenchyma, the pleura, and, to a lesser extent, the airways and the mediastinum . Lateral decubitus views are often useful for determining whether pleural abnormalities represent freely flowing fluid, whereas apical lordotic views can often visualize disease at the lung apices better than the standard PA view. Portable equipment is often used for acutely ill patients who either cannot be transported to a radiology suite or cannot stand for PA and lateral views. Portable films are more difficult to interpret owing to several limitations: (1) the single antero posterior (AP) projection obtained; (2) variability in over- and underexposure of film; (3) a shorter focal spot-film distance leading to lack of edge sharpness, and loss of fine detail; and (4) magnification of the cardiac silhouette and other anterior structures by the AP projection.
Advances in computer technology and the availability of reusable radiation detectors have allowed the development of digital or computed radiography. The images obtained in this format can be subjected to significant postprocessing analysis to improve diagnostic information. In addition, the benefit of immediate availability of the images, the ability to store images electronically, and the facility of transfer within or between health care systems have led many hospital systems to convert to digital systems.
Computed Tomography
Computed tomography (CT) offers several advantages over routine chest radiography (Figs. C-1A, B and C-2A, B). First, the use of cross-sectional images allows distinction between densities that would be superimposed on plain radiographs. Second, CT is far better than routine radiographic studies at characterizing tissue density, distinguishing subtle density differences between adjacent structures, and providing accurate size assessment of lesions.
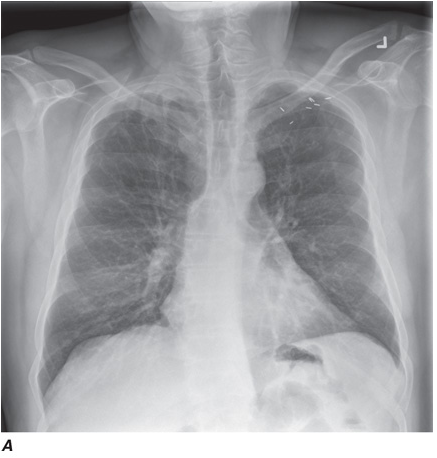
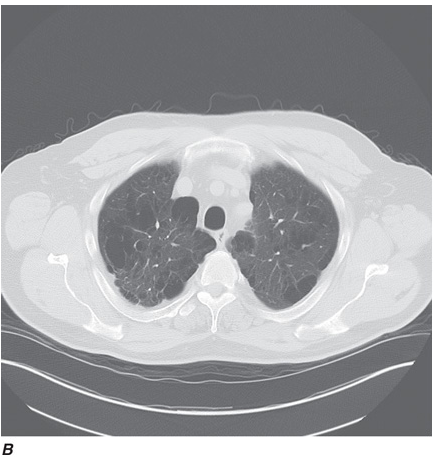
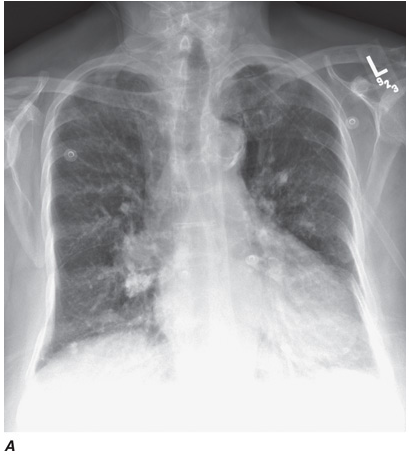
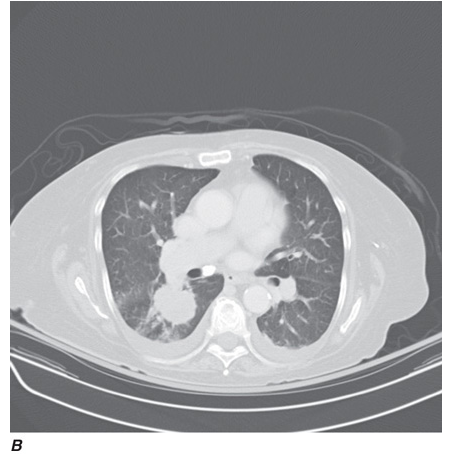
CT is particularly valuable in assessing hilar and mediastinal disease (which is often poorly characterized by plain radiography), in identifying and characterizing disease adjacent to the chest wall or spine (including pleural disease), and in identifying areas of fat density or calcification in pulmonary nodules (Figs. C-2A, B). Its utility in the assessment of mediastinal disease has made CT an important tool in the staging of lung cancer, as an assessment of tumor involvement of mediastinal lymph nodes is critical to proper staging. With the additional use of contrast material, CT also makes it possible to distinguish vascular from nonvascular structures, which is particularly important in distinguishing lymph nodes and masses from vascular structures primarily in the mediastinum, and vascular disorders such as pulmonary embolism.
In high-resolution CT (HRCT), the thickness of individual cross-sectional images is ~1–2 mm, rather than the usual 7–10 mm in conventional CT. The visible detail on HRCT scans allows better recognition of subtle parenchymal and airway disease, thickened interlobular septa, ground-glass opacification, small nodules, and the abnormally thickened or dilated airways seen in bronchiectasis. Using HRCT, characteristic patterns are recognized for many interstitial lung diseases such as lymphangitic carcinoma, idiopathic pulmonary fibrosis, sarcoidosis, and eosinophilic granuloma. However, there is debate about, the settings in which the presence of a characteristic pattern on HRCT eliminates the need for obtaining lung tissue to make a diagnosis.
Helical CT Scanning
Recent advances in computer processing have allowed the development of helical CT scanning. Helical CT technology results in faster scans with improved contrast enhancement and thinner collimation. The image is obtained during a single breath-holding maneuver that allows less motion artifact. In addition, helical CT scanning allows the collection of continuous data over a larger volume of lung than is possible with conventional CT. Data from the imaging procedure can be reconstructed as images in planes other than the traditional cross-sectional (axial) view, including coronal, or sagittal planes (Fig. C-3A). Finally, sophisticated volumetric “3D” representations of structures can be produced (Fig. C-3B) including the ability to perform a virtual bronchoscopy, mimicking direct visualization through a bronchoscope (Fig. C-4).
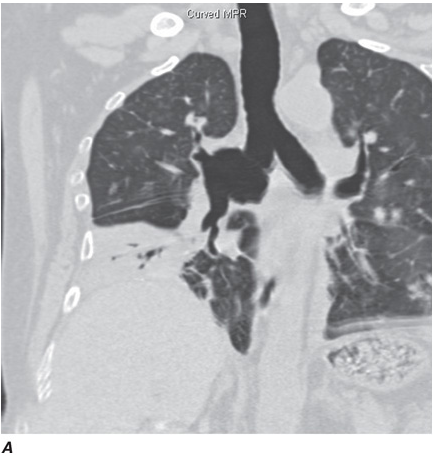
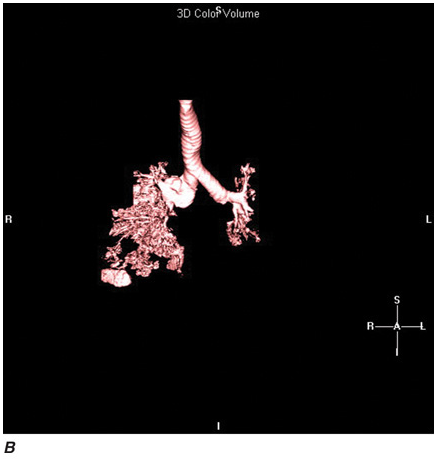

Multidetector CT (MDCT)
Refinements in detector technology have allowed production of scanners with additional detectors along the scanning axis (z-axis). These scanners, called multidetector CT (MDCT) scanners, can obtain multiple slices in a single rotation that are thinner and can be acquired in a shorter period of time. This results in enhanced resolution and increased image reconstruction ability. As the technology has progressed, higher numbers (2, 4, 6, 8, 10, 16, 32, 40 and currently up to 64) of detectors are used to produce clearer final images. The development of MDCT allows for even shorter breath holds, which are beneficial for all patients but especially children, the elderly, and the critically ill. However, it should be noted that despite the advantages of MDCT, there is an increase in radiation dose compared to single-detector CT to consider. With MDCT, the additional detectors along the z-axis result in improved use of the contrast bolus. In addition, the shorter breath holds secondary to faster scanning times and increased resolution have all led to improved imaging of the pulmonary vasculature and the ability to detect segmental and subsegmental emboli. In contrast to pulmonary angiography, CT pulmonary angiography (CTPA) also allows simultaneous detection of parenchymal abnormalities that may be contributing to a patient’s clinical presentation. Secondary to these advantages and increasing availability, CTPA has rapidly become the test of choice for many clinicians in the evaluation of pulmonary embolism; it is considered equal to pulmonary angiography in terms of accuracy, and with less associated risks.
Magnetic Resonance Imaging
The role of magnetic resonance (MR) imaging in the evaluation of respiratory system disease is less well-defined than that of CT. Magnetic resonance provides poorer spatial resolution and less detail of the pulmonary parenchyma, and for these reasons is currently not considered a substitute for CT in imaging the thorax. However, the use of hyperpolarized gas in conjunction with MR has led to the investigational use of MR for imaging the lungs, particularly in obstructive lung disease. Of note, MR examinations are difficult to obtain among several subgroups of patients. Patients who cannot lie still or who cannot lie on their backs may have MR images that are of poor quality; some tests require patients to hold their breaths for 15 to 25 seconds at a time in order to get good MR images. MR is generally avoided in unstable and/or ventilated patients and those with severe trauma because of the hazards of the MR environment and the difficulties in monitoring patients within the MR room. The presence of metallic foreign bodies, pacemakers, and intracranial aneurysm clips also preclude use of MR.
An advantage of MR is the use of nonionizing electromagnetic radiation. Additionally, MR is well suited to distinguish vascular from nonvascular structures without the need for contrast. Blood vessels appear as hollow tubular structures because flowing blood does not produce a signal on MR imaging. Therefore, MR can be useful in demonstrating pulmonary emboli, defining aortic lesions such as aneurysms or dissection, or other vascular abnormalities (Fig. C-5) if radiation and IV contrast medium cannot be used. Gadolinium can be used as an intravascular contrast agent for MR angiography (MRA); however, synchronization of data acquisition with the peak arterial bolus is one of the major challenges of MRA; the flow of contrast medium from the peripheral injection site to the vessel of interest is affected by a number of factors including heart rate, stroke volume, and the presence of proximal stenotic lesions.
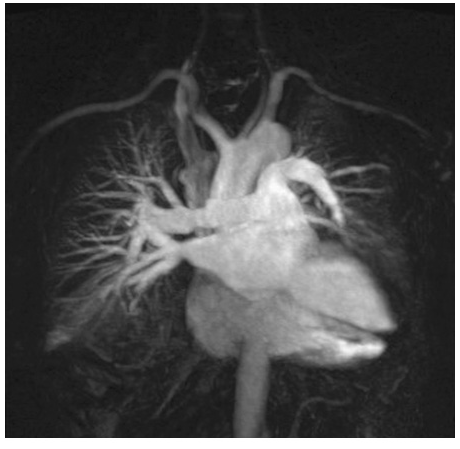
Nuclear Medicine Techniques
Nuclear imaging depends on the selective uptake of various compounds by organs of the body. In thoracic imaging, these compounds are concentrated by one of three mechanisms: blood pool or compartmentalization (e.g., within the heart), physiologic incorporation (e.g., bone or thyroid) and capillary blockage (e.g., lung scan). Radioactive isotopes can be administered by either the IV or inhaled routes or both. When injected intravenously, albumin macroaggregates labeled with [99mTc ] become lodged in pulmonary capillaries; the distribution of the trapped radioisotope follows the distribution of blood flow. When inhaled, radiolabeled xenon gas can be used to demonstrate the distribution of ventilation. Using these techniques, ventilation-perfusion lung scanning was a commonly used technique for the evaluation of pulmonary embolism. Pulmonary thromboembolism produces one or more regions of ventilation-perfusion mismatch [i.e., regions in which there is a defect in perfusion that follows the distribution of a vessel and that is not accompanied by a corresponding defect in ventilation ]. However, with advances in CT scanning, scintigraphic imaging has been largely replaced by CT angiography in patients with suspected pulmonary embolism.
Another common use of ventilation-perfusion scans is in patients with impaired lung function, who are being considered for lung resection. Because many patients with bronchogenic carcinoma have coexisting chronic obstructive pulmonary disease (COPD), the question arises as to whether or not a patient can tolerate lung resection. The distribution of the isotope(s) can be used to assess the regional distribution of blood flow and ventilation, allowing the physician to estimate the level of postoperative lung function.
Positron Emission Tomographic Scanning
Positron emission tomographic (PET) scanning is commonly used to identify malignant lesions in the lung, based on their increased uptake and metabolism of glucose. The technique involves injection of a radiolabeled glucose analogue, [18F]-fluoro-2-deoxyglucose (FDG), which is taken up by metabolically active malignant cells. However, FDG is trapped within the cell following phosphorylation, and the unstable [18F] decays by emission of positrons, which can be detected by a specialized PET camera or by a gamma camera that has been adapted for imaging of positron-emitting nuclides. This technique has been used in the evaluation of solitary pulmonary nodules and in staging lung cancer through the detection or exclusion of mediastinal lymph node involvement and identification of extrathoracic disease. The limited anatomical definition of radionuclide imaging has been improved by the development of hybrid imaging that allows the superimposition of nuclear medicine and CT images, a technique known as functional–anatomical mapping. Today, most PET scans are performed using instruments with combined PET and CT scanners. The hybrid PET/CT scans provide images that help pinpoint the abnormal metabolic activity to anatomical structures seen on CT. The combined scans provide more accurate diagnoses than the two scans performed separately. FDG–PET can differentiate benign from malignant lesions as small as 1 cm. However, false-negative findings can occur in lesions with low metabolic activity such as carcinoid tumors and bronchioloalveolar cell carcinomas, or in lesions <1 cm in which the required threshold of metabolically active malignant cells is not present for PET diagnosis. False-positive results can be seen due to FDG uptake in inflammatory conditions such as pneumonia and granulomatous diseases.
Pulmonary Angiography
The pulmonary arterial system can be visualized by pulmonary angiography, in which radiopaque contrast medium is injected through a catheter placed in the pulmonary artery. When performed in cases of pulmonary embolism, pulmonary angiography demonstrates the consequences of an intravascular thrombus—either a defect in the lumen of a vessel (a filling defect) or an abrupt termination (cutoff) of the vessel. Other, less common indications for pulmonary angiography include visualization of a suspected pulmonary arteriovenous malformation and assessment of pulmonary arterial invasion by a neoplasm. The risks associated with modern arteriography are extremely small, generally of greatest concern in patients with severe pulmonary hypertension. With advances in CT scanning, MDCT angiography (MDCTA) is replacing conventional angiography for the diagnosis of pulmonary embolism.
Ultrasound
Diagnostic ultrasound (US) produces images using echoes or reflection of the ultrasound beam from interfaces between tissues with differing acoustic properties. US is nonionizing and safe to perform on pregnant patients and children. It is helpful in the detection and localization of pleural abnormalities, and a quick and effective way of guiding percutaneous needle biopsy of peripheral lung, pleural, or chest wall lesions. US is also helpful in identifying septations within loculated collections and can facilitate placement of a needle for sampling of pleural liquid (i.e., for thoracentesis), improving the yield and safety of the procedure. Bedside availability makes it valuable in the intensive care setting. Real-time imaging can be used to assess the movement of the diaphragm. Using the Doppler mode, patterns of blood flow in both large and small vessels can be visualized. Because US energy is rapidly dissipated in air, it is not useful for evaluation of the pulmonary parenchyma and cannot be used if there is any aerated lung between the US probe and the abnormality of interest.
Endobronchial US, in which the US probe is passed through a bronchoscope, is emerging as a valuable adjunct to bronchoscopy, allowing identification and localization of pathology adjacent to airway walls or within the mediastinum, discussed further below.
Virtual Bronchoscopy
The three-dimensional (3D) image of the thorax obtained by MDCT can be digitally stored, reanalyzed, and displayed as 3D reconstructions of the airways down to the sixth- to seventh-generation. Using these computed generated reconstructions, a “virtual” bronchoscopy can be performed (Fig. C-5). Virtual bronchoscopy has been proposed as an adjunct to conventional bronchoscopy in several clinical situations: It can allow accurate assessment of the extent and length of an airway stenosis, including the airway distal to the narrowing; it can provide useful information about the relationship of the airway abnormality to adjacent mediastinal structures; and it allows preprocedure planning for therapeutic bronchoscopy to help ensure the appropriate equipment is available for the procedure. Virtual bronchoscopy can also be used to perform noninvasive follow-up of patients with treated airway lesions. Navigational systems using virtual bronchoscopy have been developed to allow pathfinding to guide the bronchoscopist to a peripheral region within the lung, allowing peripheral lung lesions to be sampled more efficiently. Finally, with the advent of endobronchial lung volume reduction surgery in the management of pulmonary emphysema, virtual bronchoscopy may be able to help target the area of peripheral lung for endobronchial valve procedures. The extent of emphysema in each segmental region together with other anatomic details may help in choosing the most appropriate subsegments. However, software packages for the generation of virtual bronchoscopic images are relatively early in development and their utilization and potential impact on patient care are still unknown. In addition to allowing virtual bronchoscopy, advances in computing capabilities and digital imaging allow the bronchoscopic images obtained through a real bronchoscopic examination to be stored as digital images and reviewed after completion of the procedure.
Medical Techniques for Obtaining Biologic Specimens
Collection of Sputum
Sputum can be collected either by spontaneous expectoration or after inhalation of an irritating aerosol such as hypertonic saline. The latter method, called sputum induction, is commonly used to obtain sputum for diagnostic studies, either because sputum is not spontaneously being produced or because of an expected higher yield of certain types of findings. Knowledge of the appearance and quality of the sputum specimen obtained is especially important when one is interested in Gram’s method and culture. Because sputum consists mainly of secretions from the tracheobronchial tree rather than the upper airway, the finding of alveolar macrophages and other inflammatory cells is consistent with a lower respiratory tract origin of the sample, whereas the presence of squamous epithelial cells in a “sputum” sample indicates contamination by secretions from the upper airways.
In addition to processing for routine bacterial pathogens by Gram’s method and culture, sputum can be processed for a variety of other pathogens, including staining and culture for mycobacteria or fungi, culture for viruses, and staining for Pneumocystis jiroveci. In the specific case of sputum obtained for evaluation of P. jiroveci pneumonia in a patient infected with HIV, for example, sputum should be collected by induction rather than spontaneous expectoration, and an immunofluorescent stain should be used to detect the organisms. Cytologic staining of sputum for malignant cells, using the traditional Papanicolaou method, allows noninvasive evaluation for suspected lung cancer. Traditional stains and cultures are now also being supplemented in some cases by immunologic techniques and by molecular biologic methods, including the use of polymerase chain reaction amplification and DNA probes.
Percutaneous Needle Aspiration (Transthoracic)
A needle can be inserted through the chest wall into a pulmonary lesion to aspirate material for analysis by cytologic or microbiologic techniques. Aspiration can be performed to obtain a diagnosis or to decompress and/or drain a fluid collection. The procedure is usually carried out under CT or ultrasound guidance to assist positioning of the needle and assure localization in the lesion. The low potential risk of this procedure (intrapulmonary bleeding or creation of a pneumothorax with collapse of the underlying lung) in experienced hands is usually acceptable owing to the information obtained. However, a limitation of the technique is sampling error due to the small size of the tissue sample. Thus, findings other than a specific cytologic or microbiologic diagnosis are of limited clinical value.
Thoracentesis
Sampling of pleural liquid by thoracentesis is commonly performed for diagnostic purposes or, in the case of a large effusion, for palliation of dyspnea. Diagnostic sampling, either by blind needle aspiration or after localization by US, allows the collection of liquid for microbiologic and cytologic studies. Analysis of the fluid obtained for its cellular composition and chemical constituents, including glucose, protein, and lactate dehydrogenase, allows the effusion to be classified as either exudative or transudative .
Bronchoscopy
Bronchoscopy is the process of direct visualization of the tracheobronchial tree. Although bronchoscopy is now performed almost exclusively with flexible fiberoptic instruments, rigid bronchoscopy, generally performed in an operating room on a patient under general anesthesia, still has a role in selected circumstances, primarily because of a larger suction channel and the fact that the patient can be ventilated through the bronchoscope channel. These situations include the retrieval of a foreign body and the suctioning of a massive hemorrhage, for which the small suction channel of the bronchoscope may be insufficient.
Flexible Fiberoptic Bronchoscopy
This outpatient procedure is usually performed in an awake but sedated patient (conscious sedation). The bronchoscope is passed through either the mouth or the nose, between the vocal cords, and into the trachea. The ability to flex the scope makes it possible to visualize virtually all airways to the level of subsegmental bronchi. The bronchoscopist is able to identify endobronchial pathology, including tumors, granulomas, bronchitis, foreign bodies, and sites of bleeding. Samples from airway lesions can be taken by several methods, including washing, brushing, and biopsy. Washing involves instillation of sterile saline through a channel of the bronchoscope and onto the surface of a lesion. A portion of the liquid is collected by suctioning through the bronchoscope, and the recovered material can be analyzed for cells (cytology) or organisms (by standard stains and cultures). Brushing or biopsy of the surface of the lesion, using a small brush or biopsy forceps at the end of a long cable inserted through a channel of the bronchoscope, allows recovery of cellular material or tissue for analysis by standard cytologic and histopathologic methods.
The bronchoscope can be used to sample material not only from the regions that can be directly visualized (i.e., the airways) but also from the more distal pulmonary parenchyma. With the bronchoscope wedged into a subsegmental airway, aliquots of sterile saline can be instilled through the scope, allowing sampling of cells and organisms even from alveolar spaces. This procedure, called bronchoalveolar lavage, has been particularly useful for the recovery of organisms such as P. jiroveci in patients with HIV infection.
Brushing and biopsy of the distal lung parenchyma can also be performed with the same instruments that are used for endobronchial sampling. These instruments can be passed through the scope into small airways, where they penetrate the airway wall, allowing biopsy of peribronchial alveolar tissue. This procedure, called transbronchial biopsy, is used when there is either relatively diffuse disease or a localized lesion of adequate size. With the aid of fluoroscopic imaging, the bronchoscopist is able to determine not only whether and when the instrument is in the area of abnormality, but also the proximity of the instrument to the pleural surface. If the forceps are too close to the pleural surface, there is a risk of violating the visceral pleura and creating a pneumothorax; the other potential complication of transbronchial biopsy is pulmonary hemorrhage. The incidence of these complications is less than several percent.
Transbronchial Needle Aspiration (TBNA)
Another procedure involves use of a hollow-bore needle passed through the bronchoscope for sampling of tissue adjacent to the trachea or a large bronchus. The needle is passed through the airway wall (transbronchial), and cellular material can be aspirated from mass lesions or enlarged lymph nodes, generally in a search for malignant cells. Other promising new techniques that are not yet widely available include fluorescence bronchoscopy (to detect early endobronchial malignancy) and endobronchial ultrasound (to better identify and localize peribronchial and mediastinal pathology). Mediastinoscopy has been considered the gold standard for mediastinal staging; however, TBNA allows sampling from the lungs and surrounding lymph nodes without the need for surgery or general anesthesia.
Endobronchial Ultrasound (EBUS)–Transbronchial Needle Aspiration (TBNA)
Further advances in needle aspiration techniques have been accomplished with the development of endobronchial ultrasound (EBUS). The technology uses an ultrasonic bronchoscope fitted with a probe that allows for needle aspiration of mediastinal and hilar lymph nodes guided by real-time US images. This procedure offers access to more difficult-to-reach areas and smaller lymph nodes in the staging of malignancies. EBUS–TBNA has the potential to access the same paratracheal and subcarinal lymph node stations as mediastinoscopy, but also extends out to the hilar lymph nodes (levels 10 and 11).The usefulness of EBUS for clinical indications other than lung cancer is unclear, although studies on sarcoidosis point to the effectiveness of endobronchial ultrasonography in diagnosing this disease.
Interventional Pulmonology (IP)
Interventional pulmonology was initially developed to focus on procedures to help palliate patients with advanced thoracic malignancies. However, the availability of advanced bronchoscopic and pleuroscopic techniques is enabling interventional pulmonologists to provide alternatives to surgery for patients with a wide variety of thoracic disorders and problems. IP can be defined as “the art and science of medicine as related to the performance of diagnostic and invasive therapeutic procedures that which require additional training and expertise beyond that which required in a standard pulmonary medicine training program.”
A central role for an IP physician is the acquisition of tissue for diagnosing mass lesions within the thorax. Several techniques already discussed are part of the day-to-day procedural armamentarium used by an IP physician. TBNA to obtain cytologic, histologic, or microbiologic sampling of lesions within the airway wall, the lung parenchyma, and mediastinum. TBNA is frequently performed in combination with EBUS to improve diagnostic yield. Transthoracic needle aspiration and biopsy (TTNA/B) refers to the percutaneous sampling of lesions involving the chest wall, lung parenchyma, and mediastinum for cytologic, histopathologic, or microbiologic examinations.
Autofluorescence Bronchoscopy
Autofluorescence bronchoscopy (AFB) uses bronchoscopes with an additional light source that allows an experienced operator (interventional pulmonologist or surgeon) to distinguish between normal and abnormal tissue. This technique can be used as a screening tool in high-risk individuals to inspect the tracheobronchial tree in order to identify premalignant lesions (airway dysplasia) and carcinoma in situ.
Medical Thoracoscopy
Medical thoracoscopy (or pleuroscopy) focuses on the diagnosis of pleural-based problems. The procedure is performed with a conventional rigid or a semirigid pleuroscope (similar in design to a bronchoscope and enabling the operator to inspect the pleural surface, sample and/or drain pleural fluid, or perform targeted biopsies of the parietal pleura). Medical thoracoscopy can be performed in the endoscopy suite or operating room with the patient under conscious sedation and local anesthesia. In contrast, video-assisted thoracoscopic surgery (VATS) requires general anesthesia and is only performed in the OR. A common diagnostic indication for medical thoracoscopy is the evaluation of a pleural effusion or biopsy of presumed parietal pleural carcinomatosis. It can also be used to place a chest tube under visual guidance, or perform chemical or talc pleurodesis, a therapeutic intervention to prevent a recurrent pleural effusion (usually malignant) or recurrent pneumothorax.
Therapeutic Bronchoscopy
The bronchoscope may provide the opportunity for treatment as well as diagnosis. A central role of the IP physican is the performance of therapeutic bronchoscopy. For example, an aspirated foreign body may be retrieved with an instrument passed through the bronchoscope (either flexible or rigid), and bleeding may be controlled with a balloon catheter similarly introduced. Newer interventional techniques performed through a bronchoscope include methods for achieving and maintaining patency of airways that are partially or completely occluded, especially by tumors. These techniques include laser therapy, cryotherapy, argon plasma coagulation, electrocautery, balloon bronchoplasty and dilation, and stent placement. Many IP physicians are also trained in performing percutaneous tracheotomy.
Surgical Techniques for Obtaining Biologic Specimens
Evaluation and diagnosis of disorders of the chest commonly involve collaboration between pulmonologists and thoracic surgeons. While procedures such as mediastinoscopy, VATS, and thoracotomy are performed by thoracic surgeons, there is overlap in many minimally invasive techniques that can be performed by a pulmonologist or a thoracic surgeon.
Mediastinoscopy and Mediastinotomy
Proper staging of lung cancer is of paramount concern when determining a treatment regimen. Although CT and PET scanning are useful for determining the size and nature of mediastinal lymph nodes as part of the staging of lung cancer, tissue biopsy and histopathologic examination are often critical for the diagnosis of mediastinal masses or enlarged mediastinal lymph nodes. The two major surgical procedures used to obtain specimens from masses or nodes in the mediastinum are mediastinoscopy (via a suprasternal approach) and mediastinotomy (via a parasternal approach). Both procedures are performed under general anesthesia by a qualified surgeon. In the case of suprasternal mediastinoscopy, a rigid mediastinoscope is inserted at the suprasternal notch and passed into the mediastinum along a pathway just anterior to the trachea. Tissue can be obtained with biopsy forceps passed through the scope, sampling masses or nodes that are in a paratracheal or pretracheal position (levels 2R, 2L, 3, 4R, 4L). Aortopulmonary lymph nodes (levels 5, 6) are not accessible by this route and thus are commonly sampled by parasternal mediastinotomy (the Chamberlain procedure). This approach involves a parasternal incision and dissection directly down to a mass or node that requires biopsy.
As an alternative to surgery, a bronchoscope can be used to perform TBNA (discussed above) to obtain tissue from the mediastinum, and, when combined with EBUS, can allow access to the same lymph node stations associated with mediastinoscopy, but also extend access out to the hilar lymph nodes (levels 10, 11). Finally, endoscopic ultrasound (EUS)–fine-needle aspiration (FNA) is a second procedure that complements EBUS–FNA in the staging of lung cancer. EUS–FNA is performed via the esophagus and is ideally suited for sampling lymph nodes in the posterior mediastinum (levels 7, 8, 9). Because US imaging cannot penetrate air filled spaces, the area directly anterior to the trachea cannot accurately be assessed and is a “blind spot” for EUS–FNA. However, EBUS–FNA can visualize the anterior lymph nodes and can complement EUS–FNA. The combination of EUS–FNA and EBUS–FNA is a technique that is becoming an alternative to surgery for staging the mediastinum in thoracic malignancies.
Video-Assisted Thoracic Surgery
Advances in video technology have allowed the development of thoracoscopy, or VATS, for the diagnosis and management of pleural as well as parenchymal lung disease. This procedure is performed in the operating room using single-lung ventilation with double-lumen endotracheal intubation and involves the passage of a rigid scope with a distal lens through a trocar inserted into the pleura. A high-quality image is shown on a monitor screen, allowing the operator to manipulate instruments passed into the pleural space through separate small intercostal incisions. With these instruments the operator can biopsy lesions of the pleura under direct visualization. In addition, this procedure is now used commonly to biopsy peripheral lung tissue or to remove peripheral nodules for both diagnostic and therapeutic purposes. This much less invasive procedure has largely supplanted the traditional “open lung biopsy” performed via thoracotomy. The decision to use a VATS technique versus performing an open thoracotomy is made by the thoracic surgeon and is based on whether a patient can tolerate the single-lung ventilation that is required to allow adequate visualization of the lung. With further advances in instrumentation and experience, VATS can be used to perform procedures previously requiring thoracotomy, including stapled lung biopsy, resection of pulmonary nodules, lobectomy, pneumonectomy, pericardial window, or other standard thoracic surgical procedures; but allows them to be performed in a minimally invasive manner.
Thoracotomy
Although frequently replaced by VATS, thoracotomy remains an option for the diagnostic sampling of lung tissue. It provides the largest amount of material, and it can be used to biopsy and/or excise lesions that are too deep or too close to vital structures for removal by VATS. The choice between VATS and thoracotomy needs to be made on a case-by-case basis.
