Bronchiectasis
Bronchiectasis refers to an irreversible airway dilation that involves the lung in either a focal or a diffuse manner and that classically has been categorized as cylindrical or tubular (the most common form), varicose, or cystic. This chapter will focus largerly on non-cyctic fibrosis bronchiectasis. The reader is referred to chapter cystic fibrosis for a more focused discussion on cystic fibrosis bronchiectasis.
Bronchiectasis can arise from infectious or noninfectious causes (Table I-1). Clues to the underlying etiology are often provided by the pattern of lung involvement. Focal bronchiectasis refers to bronchiectatic changes in a localized area of the lung and can be a consequence of obstruction of the airway—either extrinsic (e.g., due to compression by adjacent lymphadenopathy or parenchymal tumor mass) or intrinsic (e.g., due to an airway tumor or aspirated foreign body, a scarred/stenotic airway, or bronchial atresia from congenital underdevelopment of the airway). Diffuse bronchiectasis is characterized by widespread bronchiectatic changes throughout the lung and often arises from an underlying systemic or infectious disease process.
Table I-1 Major Etiologies of Bronchiectasis and Proposed Workup
| Pattern of Lung Involvement by Bronchiectasis | Etiology by Categories (with Specific Examples) | Workup |
| Focal | Obstruction (e.g., aspirated foreign body, tumor mass) | Chest imaging (chest x-ray and/or chest CT); bronchoscopy |
| Diffuse | Infection (e.g., bacterial, nontuberculous mycobacterial) | Gram’s stain/culture; stains/cultures for acid-fast bacilli and fungi. If no pathogen is identified, consider bronchoscopy with bronchoalveolar lavage (BAL) |
| Immunodeficiency (e.g., hypogammaglobulinemia, HIV infection, bronchiolitis obliterans after lung transplantation) | Complete blood count with differential; immunoglobulin measurement; HIV testing | |
| Genetic causes (e.g., cystic fibrosis, Kartagener’s syndrome, alpha1 antitrypsin deficiency) | Measurement of chloride levels in sweat (for cystic fibrosis), alpha1 antitrypsin levels; nasal or respiratory tract brush/biopsy (for dyskinetic/immotile cilia syndrome); genetic testing | |
| Autoimmune or rheumatologic causes (e.g., rheumatoid arthritis, Sjögren’s syndrome, inflammatory bowel disease); immune-mediated disease (e.g., allergic bronchopulmonary aspergillosis) | Clinical examination with careful joint exam, serologic testing (e.g., for rheumatoid factor). Consider workup for allergic bronchopulmonary aspergillosis, especially in patients with refractory asthmaa | |
| Recurrent aspiration | Test of swallowing function and general neuromuscular strength | |
| Miscellaneous (e.g., yellow nail syndrome; traction bronchiectasis from postradiation fibrosis or idiopathic pulmonary fibrosis) | Guided by clinical condition | |
| Idiopathic | Exclusion of other causes |
More pronounced involvement of the upper lung fields is most common in cystic fibrosis (CF) and is also observed in postradiation fibrosis, corresponding to the lung region encompassed by the radiation port. Bronchiectasis with predominant involvement of the lower lung fields usually has its source in chronic recurrent aspiration (e.g., due to esophageal motility disorders like those in scleroderma), end-stage fibrotic lung disease (e.g., traction bronchiectasis from idiopathic pulmonary fibrosis), or recurrent immunodeficiency-associated infections (e.g., hypogammaglobulinemia). Bronchiectasis resulting from infection by nontuberculous mycobacteria [NTM; most commonly the Mycobacterium avium–intracellulare complex (MAC)] often preferentially affects the midlung fields. Congenital causes of bronchiectasis with predominant midlung field involvement include the dyskinetic/immotile cilia syndrome. Finally, predominant involvement of the central airways is reported in association with allergic bronchopulmonary aspergillosis (ABPA), in which an immune-mediated reaction to Aspergillus damages the bronchial wall. Congenital causes of central airway–predominant bronchiectasis resulting from cartilage deficiency include tracheobronchomegaly (Mounier-Kuhn syndrome) and Williams-Campbell syndrome.
In many cases, the etiology of bronchiectasis is not determined. In case series, as many as 25–50% of patients referred for bronchiectasis have idiopathic disease.
Epidemiology
The epidemiology of bronchiectasis varies greatly with the underlying etiology. For example, patients born with CF often develop significant clinical bronchiectasis in late adolescence or early adulthood, although atypical presentations of CF in adults in their thirties and forties are also possible. In contrast, bronchiectasis resulting from MAC infection classically affects nonsmoking women older than age 50 years. In general, the incidence of bronchiectasis increases with age. Bronchiectasis is more common among women than among men.
In areas where tuberculosis is prevalent, bronchiectasis more frequently occurs as a sequela of granulomatous infection. Focal bronchiectasis can arise from extrinsic compression of the airway by enlarged granulomatous lymph nodes and/or from development of intrinsic obstruction as a result of erosion of a calcified lymph node through the airway wall (e.g., broncholithiasis). Especially in reactivated tuberculosis, parenchymal destruction from infection can result in areas of more diffuse bronchiectasis. Apart from cases associated with tuberculosis, an increased incidence of non-CF bronchiectasis with an unclear underlying mechanism has been reported as a significant problem in developing nations. It has been suggested that the high incidence of malnutrition in certain areas may predispose to immune dysfunction and development of bronchiectasis.
Pathogenesis and Pathology
The most widely cited mechanism of infectious bronchiectasis is the “vicious cycle hypothesis,” in which susceptibility to infection and poor mucociliary clearance result in microbial colonization of the bronchial tree. Some organisms, such as Pseudomonas aeruginosa, exhibit a particular propensity for colonizing damaged airways and evading host defense mechanisms. Impaired mucociliary clearance can result from inherited conditions such as CF or dyskinetic cilia syndrome, and it has been proposed that a single severe infection (e.g., pneumonia caused by Bordetella pertussis or Mycoplasma pneumoniae) can result in significant airway damage and poor secretion clearance. The presence of the microbes incites continued chronic inflammation, with consequent damage to the airway wall, continued impairment of secretion and microbial clearance, and ongoing propagation of the infectious/inflammatory cycle. Moreover, it has been proposed that mediators released directly from bacteria can interfere with mucociliary clearance.
Classic studies of the pathology of bronchiectasis from the 1950s demonstrated significant small-airway wall inflammation and larger-airway wall destruction as well as dilation, with loss of elastin, smooth muscle, and cartilage. It has been proposed that inflammatory cells in the small airways release proteases and other mediators, such as reactive oxygen species and proinflammatory cytokines, that damage the larger-airway walls. Furthermore, the ongoing inflammatory process in the smaller airways results in airflow obstruction. It is believed that antiproteases, such as alpha1 antitrypsin, play an important role in neutralizing the damaging effects of neutrophil elastase and in enhancing bacterial killing. In addition to emphysema, bronchiectasis has been observed in patients with alpha1 antitrypsin deficiency.
Proposed mechanisms for noninfectious bronchiectasis include immune-mediated reactions that damage the bronchial wall (e.g., those associated with systemic autoimmune conditions such as Sjögren’s syndrome and rheumatoid arthritis). Traction bronchiectasis refers to dilated airways arising from parenchymal distortion as a result of lung fibrosis (e.g., postradiation fibrosis or idiopathic pulmonary fibrosis).
Clinical Manifestations
The most common clinical presentation is a persistent productive cough with ongoing production of thick, tenacious sputum. Physical findings often include crackles and wheezing on lung auscultation, and some patients with bronchiectasis exhibit clubbing of the digits. Mild to moderate airflow obstruction is often detected on pulmonary function tests, overlapping with that seen at presentation with other conditions, such as chronic obstructive pulmonary disease (COPD). Acute exacerbations of bronchiectasis are usually characterized by changes in the nature of sputum production, with increased volume and purulence. However, typical signs and symptoms of lung infection, such as fever and new infiltrates, may not be present.
Diagnosis
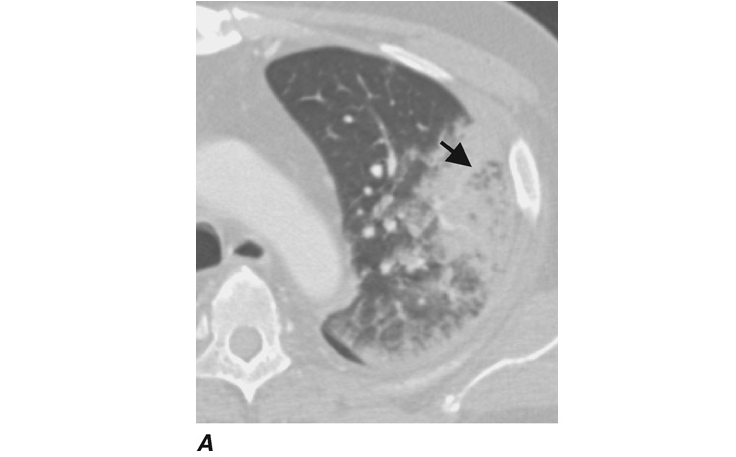
The diagnosis is usually based on presentation with a persistent chronic cough and sputum production accompanied by consistent radiographic features. While chest radiographs lack sensitivity, the presence of “tram tracks” indicating dilated airways is consistent with bronchiectasis. Chest CT is more specific for bronchiectasis and is the imaging modality of choice for confirming the diagnosis. CT findings include airway dilation (detected as parallel “tram tracks” or as the “signet-ring sign”—a cross-sectional area of the airway with a diameter at least 1.5 times that of the adjacent vessel), lack of bronchial tapering (including the presence of tubular structures within 1 cm from the pleural surface), bronchial wall thickening in dilated airways, inspissated secretions (e.g., the “tree-in-bud” pattern), or cysts emanating from the bronchial wall (especially pronounced in cystic bronchiectasis; Fig. I-1).

Approach to the Patient: Bronchiectasis
The evaluation of a patient with bronchiectasis entails elicitation of a clinical history, chest imaging, and a workup to determine the underlying etiology. Evaluation of focal bronchiectasis almost always requires bronchoscopy to exclude airway obstruction by an underlying mass or foreign body. A workup for diffuse bronchiectasis includes analysis for the major etiologies (Table I-1). Pulmonary function testing is an important component of a functional assessment of the patient.
Treatment: Bronchiectasis
Treatment of infectious bronchiectasis is directed at the control of active infection and improvements in secretion clearance and bronchial hygiene so as to decrease the microbial load within the airways and minimize the risk of repeated infections.
Antibiotic Treatment
Antibiotics targeting the causative or presumptive pathogen (with Haemophilus influenzae and P. aeruginosa isolated commonly) should be administered in acute exacerbations, usually for a minimum of 7–10 days. Decisions about treatment of NTM infection can be difficult, given that these organisms can be colonizers as well as pathogens and the prolonged treatment course often is not well tolerated. Consensus guidelines have advised that diagnostic criteria for true clinical infection with NTM should be considered in patients with symptoms and radiographic findings of lung disease who have at least two sputum samples positive on culture; at least one bronchoalveolar lavage (BAL) fluid sample positive on culture; a biopsy sample displaying histopathologic features of NTM infection (e.g., granuloma or a positive stain for acid-fast bacilli) along with one positive sputum culture; or a pleural fluid sample (or a sample from another sterile extrapulmonary site) positive on culture. MAC strains are the most common NTM pathogens, and the recommended regimen for HIV-negative patients includes a macrolide combined with rifampin and ethambutol. Consensus guidelines also recommend macrolide susceptibility testing for clinically significant MAC isolates.
Bronchial Hygiene
The numerous approaches employed to enhance secretion clearance in bronchiectasis include hydration and mucolytic administration, aerosolization of bronchodilators and hyperosmolar agents (e.g., hypertonic saline), and chest physiotherapy (e.g., postural drainage, traditional mechanical chest percussion via hand clapping to the chest, or use of devices such as an oscillatory positive expiratory pressure flutter valve or a high-frequency chest wall oscillation vest). The mucolytic dornase (DNase) is recommended routinely in CF-related bronchiectasis but not in non-CF bronchiectasis, given concerns about lack of efficacy and potential harm in the non-CF population.
Anti-Inflammatory Therapy
It has been proposed that control of the inflammatory response may be of benefit in bronchiectasis, and relatively small-scale trials have yielded evidence of alleviated dyspnea, decreased need for inhaled B-agonists, and reduced sputum production with inhaled glucocorticoids. However, no significant differences in lung function or bronchiectasis exacerbation rates have been observed. Risks of immunosuppression and adrenal suppression must be carefully considered with use of anti-inflammatory therapy in infectious bronchiectasis. Nevertheless, administration of oral/systemic glucocorticoids may be important in treating bronchiectasis due to certain etiologies, such as ABPA, or noninfectious bronchiectasis due to underlying conditions, especially that in which an autoimmune condition is believed to be active (e.g., rheumatoid arthritis or Sjögren’s syndrome). Patients with ABPA may also benefit from a prolonged course of treatment with the oral antifungal agent itraconazole.
Refractory Cases
In select cases, surgery can be considered, with resection of a focal area of suppuration. In advanced cases, lung transplantation can be considered.
Complications
In more severe cases of infectious bronchiectasis, recurrent infections and repeated courses of antibiotics can lead to microbial resistance to antibiotics. In certain cases, combinations of antibiotics that have their own independent toxicity profiles may be necessary to treat resistant organisms.
Recurrent infections can result in injury to superficial mucosal vessels, with bleeding and, in severe cases, life-threatening hemoptysis. Management of massive hemoptysis usually requires intubation to stabilize the patient, identifying the source of bleeding, and protecting the nonbleeding lung. Control of bleeding often necessitates bronchial artery embolization and, in severe cases, surgery.
Prognosis
Outcomes of bronchiectasis vary widely with the underlying etiology and may also be influenced by the frequency of exacerbations and (in infectious cases) the specific pathogens involved. In one study, the decline of lung function in patients with non-CF bronchiectasis was similar to that in patients with COPD, with the forced expiratory volume in 1 s (FEV1) declining by 50–55 mL per year as opposed to 20–30 mL per year for healthy controls.
Prevention
Reversal of an underlying immunodeficient state (e.g., by administration of gamma globulin for immunoglobulin-deficient patients) and vaccination of patients with chronic respiratory conditions (e.g., influenza and pneumococcal vaccines) can decrease the risk of recurrent infections. Patients who smoke should be counseled about smoking cessation.
After resolution of an acute infection in patients with recurrences (e.g., ≥3 episodes per year), the use of suppressive antibiotics to minimize the microbial load and reduce the frequency of exacerbations has been proposed, although there is less consensus with regard to this approach in non-CF-associated bronchiectasis than there is in patients with CF-related bronchiectasis. Possible suppressive treatments include (1) administration of an oral antibiotic (e.g., ciprofloxacin) daily for 1–2 weeks per month; (2) use of a rotating schedule of oral antibiotics (to minimize the risk of development of drug resistance); (3) administration of a macrolide antibiotic daily or three times per week (with mechanisms of possible benefit related to non-antimicrobial properties, such as anti-inflammatory effects and reduction of gram-negative bacillary biofilms); (4) inhalation of aerosolized antibiotics [e.g., tobramycin inhalation solution (TOBI)] by select patients on a rotating schedule (e.g., 30 days on, 30 days off) with the goal of decreasing the microbial load without encountering the side effects of systemic drug administration; and (5) intermittent administration of IV antibiotics (e.g., “clean-outs”) for patients with more severe bronchiectasis and/or resistant pathogens.
In addition, ongoing, consistent attention to bronchial hygiene can promote secretion clearance and decrease the microbial load in the airways.
Lung Abscess
The term lung abscess refers to a microbial infection of the lung that results in necrosis of the pulmonary parenchyma. Necrotizing pneumonia or lung gangrene refers to multiple small pulmonary abscesses in contiguous areas of the lung, usually resulting from a more virulent infection.
Classification
Lung abscesses are classified by clinical and pathologic features including the tempo of progression, the presence or absence of an associated underlying lesion, and the microbial pathogen responsible. Duration defines the infection as acute versus chronic, with the dividing line usually at 4–6 weeks. Abscesses occurring in the presence of underlying pulmonary lesions, including tumors or systemic conditions (e.g., HIV infection), are referred to as secondary; those that occur in the absence of underlying pulmonary lesions are considered primary. The term nonspecific lung abscess refers to cases in which no likely pathogen is recovered from expectorated sputum; most such cases are presumed to be due to anaerobic bacteria. Putrid lung abscess is a term applied to anaerobic bacterial lung abscesses, which are characterized by distinctive foul-smelling breath, sputum, or empyema fluid.
Etiology
The likely etiologic agent, appropriate diagnostic testing, and appropriate treatment are frequently indicated by the characteristics of the host and the disease process. A variety of microbial pathogens cause lung abscess (Table I-2). Most nonspecific lung abscesses are presumed to be due to anaerobic bacteria. Mycobacteria, especially M. tuberculosis, are a very important cause of pulmonary infections and abscess formation. Fungi and some parasites also cause lung abscess. An acute lung abscess developing in a young, previously healthy patient, especially in conjunction with influenza, is likely to involve Staphylococcus aureus; this pathogen generally is seen easily on sputum Gram’s stain and culture, and presumptive treatment for methicillin-resistant S. aureus is urgent. In an immunocompromised host, suspected pathogens include enteric gram-negative bacilli—especially Klebsiella pneumoniae but also agents that are found almost exclusively in patients with defective cell-mediated immunity, such as Nocardia asteroides and Rhodococcus equi. Lung abscess acquired in other countries may involve Burkholderia pseudomallei or Paragonimus westermani.
Table I-2 Microbial Pathogens Causing Cavitary Lung Infection
| Aspiration-Prone Host |
| Anaerobic bacteria plus microaerophilic and/or anaerobic streptococci, Gemella spp. |
| Embolic (endovascular) lesions: usually Staphylococcus aureus, Pseudomonas aeruginosa, Fusobacterium necrophoruma |
| Endemic fungi: Histoplasma, Blastomyces, Coccidioides spp. |
| Mycobacteria: M. tuberculosis, M. kansasii, M. avium |
| Immunocompromised Host |
| M. tuberculosis, Nocardia asteroides, Rhodococcus equi, Legionella spp., P. aeruginosa, Enterobacteriaceae (especially Klebsiella pneumoniae), Aspergillus spp., Cryptococcus spp. |
| Previously Healthy Host |
| Bacteria: S. aureus,bS. milleri, K. pneumoniae, group A Streptococcus; Gemella, Legionella, and Actinomyces spp. |
| Parasites: Entamoeba histolytica, Paragonimus westermani, Strongyloides stercoralis |
bOften in a young patient with influenza.
Multiple pulmonary lesions that are not caused by microbes may resemble lung abscess. These include the lesions of pulmonary infarction, bronchiectasis, necrotizing carcinoma, pulmonary sequestration, vasculitides [e.g., periarteritis nodosa, granulomatosis with polyangiitis (Wegener’s), Goodpasture syndrome], and cysts or bullae with fluid collections. In some cases, multiple lung abscesses result from septic emboli, most commonly in association with tricuspid valve endocarditis.
Clinical Features
The classic presentation of nonspecific lung abscess is an indolent infection that evolves over several days or weeks, usually in a host who has a predisposition to aspiration. A common feature is periodontal infection with pyorrhea or gingivitis. Anaerobes and aerobic or microaerophilic streptococci that colonize the upper airways are implicated in these lesions. The usual symptoms are fatigue, cough, sputum production, and fever. Chills are uncommon. Many patients have evidence of chronic disease, such as weight loss and anemia. Some patients have putrid-smelling sputum indicative of the presence of anaerobes; the foul odor is presumably due to the organisms’ production of short-chain fatty acids, such as butyric or succinic acid. Some patients have pleurisy due to pleural involvement by contiguous spread or by a bronchopleural fistula. The pleurisy may be severe and may be the symptom that prompts medical evaluation. Sequential x-rays or CT scans show the evolution of this lesion from pneumonitis to cavitation, a process that generally requires 7–14 days in experimental animals (Fig. I-2).

Diagnosis
Lung abscess can usually be detected with standard imaging, including chest x-ray and CT (Fig. I-2). The latter is clearly preferred for precise definition of the lesion and its location and possibly for detection of underlying lesions. Lymphadenopathy is not associated with bacterial lung abscess; thus this finding suggests an alternative diagnosis.
Microbiologic studies include stains and cultures of expectorated sputum to detect aerobic bacterial pathogens. However, clinical correlations are very important because sputum cultures (especially those that do not satisfy standard cytologic criteria) are unreliable. In appropriate settings, it is important to consider cultures for fungi and mycobacteria. Anaerobic bacteria, the most common causes of primary lung abscess, are not detected in expectorated sputum cultures, and in any case the specimen is subject to anaerobic contamination as it traverses the upper airways. Alternative specimens that may be useful include pleural fluid obtained by thoracentesis in patients who have empyema and quantitative bronchoalveolar lavage (BAL) specimens if they are processed promptly and appropriately for anaerobic bacteria. Many reports describe the use of transtracheal aspiration to bypass the upper airways and obtain a specimen for meaningful anaerobic culture. This procedure, which was used extensively in the 1970s, has largely been abandoned out of concern about adverse consequences and because of a general decline in the pursuit of an etiologic agent in pulmonary infections. Another invasive method for bypassing contamination by the flora of the upper airways is transthoracic needle aspiration under CT guidance; the popularity of this procedure has increased in recent years. In most cases, the etiology of anaerobic lung abscess is clear: the host is prone to aspiration and has an abscess in a dependent pulmonary segment, with no other likely cause. As stated above, putrid breath, sputum, or empyema fluid indicates anaerobic infection.
Treatment: Lung Abscess
Antibiotic Selection
Treatment depends on the presumed or established etiology. Infections caused by anaerobic bacteria should usually be treated with clindamycin; the initial IV dosage of 600 mg four times daily can be changed to an oral dosage of 300 mg four times daily once the patient becomes afebrile and improves clinically. The duration of therapy is arbitrary, but many experts recommend continuation of oral treatment until imaging shows that chest lesions have cleared or have left a small, stable scar. A shorter course may be effective. An alternative to clindamycin is any B-lactam/B-lactamase inhibitor combination; parenteral treatment may be followed by orally administered amoxicillin/clavulanate. Carbapenems are also effective against anaerobic bacteria as well as streptococci, but the published data with these drugs in the treatment of anaerobic pulmonary infections are sparse. Penicillin was previously regarded as a preferred drug for these infections, but many oral anaerobes produce B-lactamases, and clindamycin proved superior to penicillin G in a randomized clinical trial. Metronidazole is highly active against virtually all anaerobes but not against aerobic microaerophilic streptococci, which play an important role in mixed infections. In therapeutic trials, metronidazole has done poorly unless combined with a B-lactam or another agent active against aerobic and microaerophilic streptococci.
Persistence of fever beyond 5–7 days or progression of the infiltrate suggests failure of therapy and a need to exclude factors such as obstruction, complicating empyema, and involvement of antibiotic-resistant bacteria. Many patients with uncomplicated lung abscesses and all those with atypical presentations or unresponsive abscesses should undergo bronchoscopy and/or CT to detect a possible associated anatomic lesion, such as a tumor, or a foreign body. Quantitative bacteriologic studies using a protected brush catheter or BAL are much less reliable when done after antibiotic therapy. Postural drainage was previously popular for patients with lung abscess, but aggressive attempts to implement this strategy may result in spillage to other pulmonary segments, leading to airway obstruction and clinical deterioration.
Lung abscess due to S. aureus is usually treated with vancomycin at a dosage that targets a trough serum level of 15–20 microgram/mL. The main alternative is linezolid. Daptomycin should not be used for pulmonary infections. Lung abscesses caused by aerobic gram-negative bacteria need to be treated according to the results of antibiotic sensitivity tests. Most common among the pathogens involved are K. pneumoniae (especially the K1 strain in Taiwan) and P. aeruginosa in patients with severe chronic lung disease or compromised immune defenses. Pseudomonal lung abscesses usually require prolonged courses of parenteral antibiotics. Carbapenems or B-lactams are frequently combined with aminoglycosides; oral fluoroquinolones are often effective initially, but resistance is common with prolonged use. Aerosolized colistin and aminoglycosides are sometimes used to augment other therapy, but the efficacy of this approach is variable.
Surgery for lung abscesses was developed at the time penicillin became available in the late 1940s. The relative roles of penicillin and resectional surgery were hotly debated at that time, but by the late 1950s penicillin was favored. Initially the standard choice for most lung abscesses, penicillin was subsequently supplanted by the options summarized above. Recent large-scale reviews indicate that, in general, surgery is now reserved for ~10–12% of patients. The major indications for surgery are failure to respond to medical management, suspected neoplasm, and hemorrhage. Failure to respond to antibiotics is usually due to an obstructed bronchus and an extremely large abscess (>6 cm in diameter) or to infection involving relatively resistant bacteria, such as P. aeruginosa. The usual procedure is lobectomy. An alternative intervention that is becoming popular is percutaneous drainage under CT guidance. Aspirate samples for assay of possible pathogens should be carefully collected.
Response to Therapy
Patients with lung abscess usually show clinical improvement, with decreased fever, within 3–5 days of initiation of antibiotic treatment. Defervescence can be expected within 5–10 days. Patients with fevers persisting for 7–14 days should undergo bronchoscopy or other diagnostic tests to better define anatomic changes and microbiologic findings. Cultures of expectorated sputum are not likely to be helpful at this juncture except for detecting pathogens such as mycobacteria and fungi. The response to therapy apparent on serial chest radiographs is delayed in comparison with the clinical course. In fact, infiltrates usually progress during the first 3 days of treatment in approximately one-half of patients. Pleural involvement is relatively common and may develop in dramatic fashion. The most common causes of failures of medical management include a failure to drain pleural collections, an inappropriate choice of antimicrobial therapy, an obstructed bronchus that prevents drainage, a “giant” abscess, a resistant pathogen, or refractory lesions due to immunocompromise.
